Cisplatin [15663-27-1]
Référence NB-64-06877-100mg
Conditionnement : 100mg
Marque : Neo Biotech
Cisplatin (Synonyms: cis-Diaminodichloroplatinum, CDDP)
Cisplatin
Copy Product InfoCisplatin (CDDP) is a chemotherapeutic agent with antitumor activity and is a classic DNA crosslinker. It inhibits DNA synthesis and induces DNA damage in cancer cells by forming DNA adducts, ultimately leading to cell death. In addition, Cisplatin can activate ferroptosis and induce autophagy. In animal studies, it is commonly used to establish models of chronic kidney injury and acute renal failure.
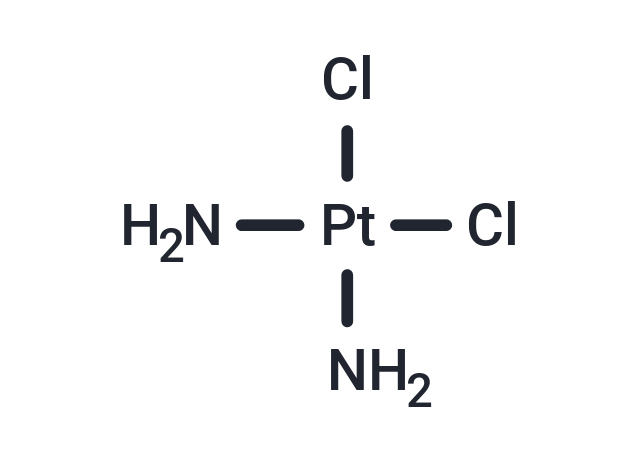
Product Introduction
| Description | Cisplatin (CDDP) is a chemotherapeutic agent with antitumor activity and is a classic DNA crosslinker. It inhibits DNA synthesis and induces DNA damage in cancer cells by forming DNA adducts, ultimately leading to cell death. In addition, Cisplatin can activate ferroptosis and induce autophagy. In animal studies, it is commonly used to establish models of chronic kidney injury and acute renal failure. |
| Targets&IC50 | SK-MES-1 cells:4.09 µM, MCF-7 cells:1.2 µM, A549 cells:1.58 µM, HCC38 cells:2.5 µM |
| In vitro | METHODS: Human non-small cell lung cancer cells A549, SKMES-1, MOR and H460 were treated with Cisplatin (0.001-100 μM) for 72 h, and cell growth inhibition was detected by MTT. RESULTS: Cisplatin dose-dependently inhibited the cell growth of A549, SKMES-1, MOR and H460 cells with IC50 of 1.58 µM, 4.09 µM, 6.39 µM and 5.72 µM, respectively. [1] METHODS: Human breast cancer MCF-7 and MDA-MB-231 were treated with Cisplatin (2-10 μg/mL) for 48 h, and the expression levels of target proteins were detected by Western Blot. RESULTS: Cisplatin dose-dependently induced increased levels of apoptosis-related proteins cleaved-caspase 3 and cleaved-PARP in MCF-7 and MDA-MB-231 cells. [2] METHODS: Mesothelioma cells JU77, LO68 and ONE58 were treated with Cisplatin (5-100 μg/mL) for 24 h, and mitochondrial membrane potential (MMP) was detected using JC-1 dye. RESULTS: Cisplatin dose-dependently decreased MMP and inhibited mitochondrial function in JU77, LO68 and ONE58 cells. [3] |
| In vivo | METHODS: To assay anti-tumor activity in vivo, Cisplatin (5 mg/kg/6 days) and Chloroquine (13 mg/kg/day) were intraperitoneally injected into BALB/c nude mice bearing hypopharyngeal squamous cell carcinoma tumors (HSCC) FaDu for eighteen days. RESULTS: Cisplatin treatment significantly inhibited HSCC tumor growth. Chloroquine inhibited autophagy and increased Cisplatin-induced apoptosis, which enhanced the efficacy of Cisplatin, resulting in reduced tumor growth and prolonged survival in mice. [4] METHODS: To attenuate nephrotoxicity induced by Cisplatin treatment, Cisplatin (3-6 mg/kg/3 days) was administered intraperitoneally and Cilastatin (100 mg/kg/day) subcutaneously to BALB/c mice with human lung adenocarcinoma tumor A549 for seven days. RESULTS: Cilastatin can reduce the induced nephrotoxicity of Cisplatin without affecting its antitumor effects. [5] |
| Disease Modeling Protocol | Nephrotoxicity model
*Precautions: They were euthanized on the 9th day of the experiment. |
| Cell Research | Rabbit renal proximal tubules were isolated using the iron oxide perfusion method and grown in 35-mm tissue culture dishes under improved conditions as described previously. The cell culture medium was a 1:1 mixture of Dulbecco's modified Eagle's medium/Ham's F-12 (without D-glucose, phenol red, or sodium pyruvate) supplemented with 15 mM HEPES buffer, 2.5 mM L-glutamine, 1 μM pyridoxine HCl, 15 mM sodium bicarbonate, and 6 mM lactate. Hydrocortisone (50 nM), selenium (5 ng/ml), human transferrin (5 μg/ml), bovine insulin (10 nM), and L-ascorbic acid-2-phosphate (50 μM) were added to fresh culture medium immediately before daily media change. In general, confluent RPTCs were treated with inhibitors or diluent control [typically DMSO at 0.1% (v/v)] for 30 min before treatment with cisplatin. Aliquots of RPTCs were used for various assays as detailed below [1]. |
| Animal Research | Mice were divided randomly into three groups (Control, Cisplatin and Cisplatin+HemoHIM), and each group consisted of twenty mice. B16F0 melanoma (5 × 10^5 cells/mouse) was inoculated into subcutaneous femoral left region of mice at 3 days before an initial injection of cisplatin. Cisplatin was injected intraperitoneally at 4 mg/kg body weight (B.W.) on day 0, 7 and 14 (total three injections). Experimental group was intubated with HemoHIM at a final concentration of 100 mg/kgB.W. by everyday from day -1 to day 16, while the control group received only water. On day 17 after initial injection of cisplatin, all mice of each group were experimented, respectively, to evaluate tumor weight or tumor size. The tumor size was calculated as follows: tumor size = ab^2/2, where a and b are the larger and smaller diameters, respectively [3]. |
| Synonyms | cis-Diaminodichloroplatinum, CDDP |
| Molecular Weight | 300.04 |
| Formula | Cl2H6N2Pt |
| Cas No. | 15663-27-1 |
| Smiles | N[Pt](Cl)(Cl)N |
| Relative Density. | 3.7 |
| Storage | Powder: -20°C for 3 years Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | H2O: 1 mg/mL (3.3 mM), Heating to 50℃ is recommended. The compound is unstable in solution. Please use soon.(DMSO can inactivate Cisplatin's activity.)  DMF: 20 mg/mL (66.66 mM), Sonication is recommended. The compound is unstable in solution. Please use soon.  | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
H2O/DMF
DMF
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||


