Atezolizumab [1380723-44-3]
Référence T9902-1mg
Conditionnement : 1mg
Marque : TargetMol
Select Batch
Purity:99.3% (SEC-HPLC)
Appearance:Liquid
Color:Transparent
Product Introduction
Bioactivity
Related Conjugates and Formulations
Antigen Details
Chemical Properties
Antibody Information
Storage & Solubility Information
| Description | Atezolizumab is an antibody inhibitor, a humanized monoclonal antibody, IgG1, which targets PD-L1 and blocks the interaction of PD-L1 with PD-1. Atezolizumab has antitumor activity and promotes T-cells to attack tumor cells. |
| In vitro | METHODS: MDA-MB-231, BT-20, MDA-MB-468 cells and T cells were co-cultured and treated with Atezolizumab (50-100 µg/mL) for 4-24 h. T cell-mediated cytotoxicity assay was performed. RESULTS: Atezolizumab significantly enhanced T cell-mediated cytotoxicity of MDA-MB-231 cells in a dose-dependent manner, with 100 µg/mL concentration of Atezolizumab showing significant efficacy at 4 and 24 h incubation.Similar results were obtained with BT-20. No Atezolizumab-induced T cell-mediated cytotoxicity was observed in PD-L1- MDA-MB-468 cells compared to MDA-MB-231 and BT-20 cells. [1] METHODS: MDA-MB-231 cells were treated with Atezolizumab (0.5 µg/mL) for 24 h. Surface expression of PD-L1 was detected by flow cytometry. RESULTS: Almost all MDA-MB-231 cells were positive for PD-L1, but the detection of PD-L1 epitopes was blocked by the specific antibody Atezolizumab after 24 h treatment. [2] |
| In vivo | METHODS: To detect in vivo antitumor activity, Atezolizumab (10 mg/kg) and Bevacizumab (5 mg/kg) were intraperitoneally injected into BALB/C nude mice bearing A2780cis xenografts every two days for three weeks. RESULTS: In vivo treatment with Atezolizumab or Bevacizumab induced significant antitumor effects and significantly inhibited tumor growth. Dual blockade with Atezolizumab and Bevacizumab significantly inhibited tumor growth compared to each treatment. [3] |
| Cell Research | Objective: Determine the binding of [111In]PD-L1-mAb to tumor cell lines. Cells: NCI-H2444(Lung Cancer cell line), MDAMB231(Breast Cancer cell line),etc. Concentrations: 1 μCi/100μl. Incubation Time: 1 h. Method: Incubating 1 μCi of [111In]PD-L1-mAb with 1×106cells (in triplicate for each cell line) for 1h at 37°C. PD-L1 blocking was performed by adding a 10-fold molar equivalent excess of the non-labeled mAb. After incubation, cells were washed three times with cold PBS prior to counting on an automated gamma counter. |
| Reactivity | Human |
| Verified Activity | Immobilized Human PD-L1 Protein (His) (TMPY-04343) at 1 μg/mL (100 μL/well) can bind Atezolizumab. The EC50 is 0.980 ng/mL. 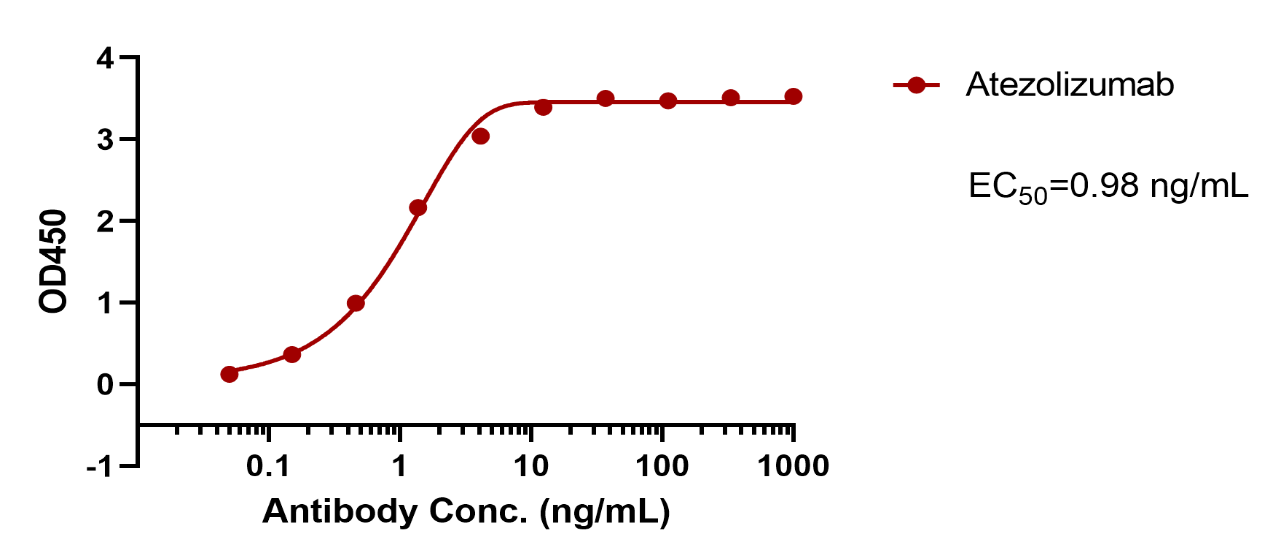 |
| Application | Functional assay |
| Antibody Type | Monoclonal |
| Formulation | Supplied as a sterile solution in a buffered formulation system (e.g., phosphate-, citrate-, or amino acid-based). Please refer to the CoA for lot-specific composition. |
| Endotoxin | <1.0 EU/mg |
| Conjucates | Unconjugated |
| Gene ID | 29126 |
| Uniprot ID | Q9NZQ7 |
| Target | B7-H1/PD-L1/CD274 |
| Molecular Weight | 144.6 kDa |
| Cas No. | 1380723-44-3 |
| Relative Density. | no data available |
| Isotype | Human IgG1 kappa |
| Recommended Isotype Control | Human IgG1 kappa, Isotype Control |
| Storage | Shipping with blue ice/Shipping at ambient temperature. |



