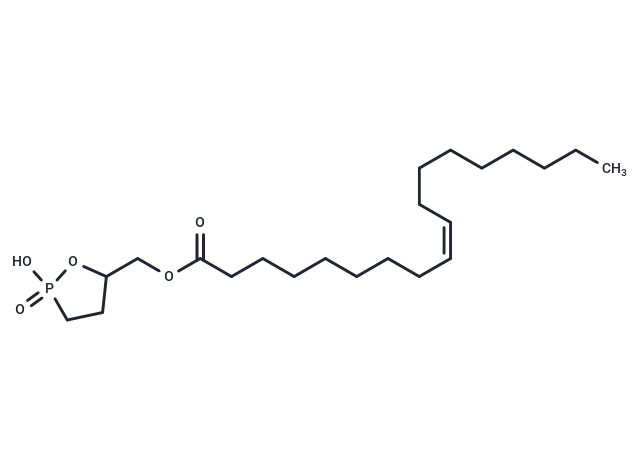
Oleoyl 3-carbacyclic Phosphatidic Acid [779333-58-3]
Katalog-Nummer NB-64-86975-50mg
Size : 50mg
Marke : Neo Biotech
Oleoyl 3-carbacyclic Phosphatidic Acid (Synonyms: 3-ccPA 18:1)
Oleoyl 3-carbacyclic Phosphatidic Acid
Copy Product InfoCyclic Phosphatidic Acids (cPAs) are naturally occurring lysophosphatidic acid (LPA) analogs, characterized by a 5-membered ring formed between the sn-2 hydroxy group and the sn-3 phosphate. Carba-derivatives of cPA (ccPA) modify the sn-2 (2-ccPA) or sn-3 (3-ccPA) linkage, hindering the conversion of cPA into LPA. Oleoyl 3-Carbacyclic Phosphatidic Acid (3-ccPA 18:1) incorporates the 18:1 fatty acid oleate at the sn-1 position on the glycerol backbone, acting as a cyclic LPA analog. This compound, at a concentration of 25 μM, blocks MM1 cells' transcellular migration through mesothelial cell monolayers induced by fetal bovine serum (by 90.1%) or LPA (by 99.9%), without impeding cell proliferation. Additionally, 3-ccPA 18:1, in the 0.1-1.0 μM range, notably suppresses autotaxin, which plays a vital role in various cancer cell behaviors including survival, growth, migration, invasion, and metastasis.

Product Introduction
| Description | Cyclic Phosphatidic Acids (cPAs) are naturally occurring lysophosphatidic acid (LPA) analogs, characterized by a 5-membered ring formed between the sn-2 hydroxy group and the sn-3 phosphate. Carba-derivatives of cPA (ccPA) modify the sn-2 (2-ccPA) or sn-3 (3-ccPA) linkage, hindering the conversion of cPA into LPA. Oleoyl 3-Carbacyclic Phosphatidic Acid (3-ccPA 18:1) incorporates the 18:1 fatty acid oleate at the sn-1 position on the glycerol backbone, acting as a cyclic LPA analog. This compound, at a concentration of 25 μM, blocks MM1 cells' transcellular migration through mesothelial cell monolayers induced by fetal bovine serum (by 90.1%) or LPA (by 99.9%), without impeding cell proliferation. Additionally, 3-ccPA 18:1, in the 0.1-1.0 μM range, notably suppresses autotaxin, which plays a vital role in various cancer cell behaviors including survival, growth, migration, invasion, and metastasis. |
| Synonyms | 3-ccPA 18:1 |
| Molecular Weight | 416.5 |
| Formula | C22H41O5P |
| Cas No. | 779333-58-3 |
| Smiles | C(OC(CCCCCCC/C=C\CCCCCCCC)=O)C1CCP(=O)(O)O1 |
| Storage | Shipping with blue ice/Shipping at ambient temperature. |


