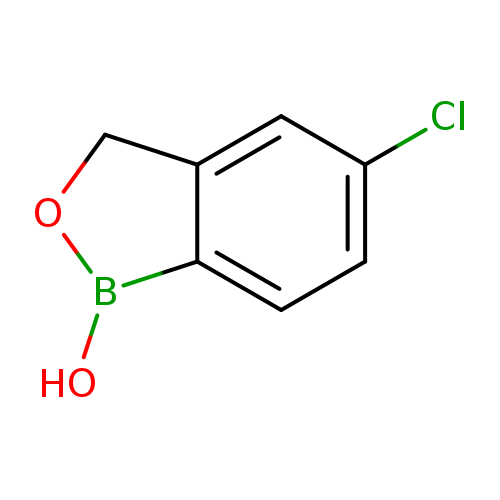
5-Chloro-1,3-dihydro-1-hydroxy-2,1-benzoxaborole [174672-06-1]
Referentie AG00AMH2-5g
Formaat : 5g
Merk : Angene
5-chloro-1,3-dihydro-2,1-benzoxaborol-1-ol
| Chemical Name: | 5-chloro-1,3-dihydro-2,1-benzoxaborol-1-ol |
| CAS Number: | 174672-06-1 |
| Product Number: | AG00AMH2(AGN-PC-0WO1M1) |
| Synonyms: | |
| MDL No: | MFCD18633195 |
| Molecular Formula: | C7H6BClO2 |
| Molecular Weight: | 168.3853 |
Identification/Properties
Computed Properties
Molecular Weight:
168.383g/mol
Hydrogen Bond Donor Count:
1
Hydrogen Bond Acceptor Count:
2
Rotatable Bond Count:
0
Exact Mass:
168.015g/mol
Monoisotopic Mass:
168.015g/mol
Topological Polar Surface Area:
29.5A^2
Heavy Atom Count:
11
Formal Charge:
0
Complexity:
155
Isotope Atom Count:
0
Defined Atom Stereocenter Count:
0
Undefined Atom Stereocenter Count:
0
Defined Bond Stereocenter Count:
0
Undefined Bond Stereocenter Count:
0
Covalently-Bonded Unit Count:
1
Compound Is Canonicalized:
Yes
Safety Information
GHS Pictogram:
N/A
Signal Word:
UN#:
N/A
Hazard Statements:
-
Precautionary Statements:
Class:
N/A
Packing Group:
N/A
NMR Spectrum
Other Analytical Data
Request for Quotation
Customer Feedback
5-Chloro-1,3-dihydro-1-hydroxy-2,1-benzoxaborole is a versatile compound that finds significant utility in chemical synthesis. In particular, it serves as a key building block in the creation of boron-containing molecules, which are valuable in a wide range of applications within the field of organic chemistry. This compound can be employed as a reactive intermediate in various reactions, including cross-coupling reactions and Suzuki-Miyaura coupling reactions, to facilitate the formation of complex organic structures. Additionally, its unique boron functionality allows for the selective modification of organic molecules, enabling the introduction of boron groups into target compounds with high precision. Overall, 5-Chloro-1,3-dihydro-1-hydroxy-2,1-benzoxaborole plays a crucial role in the synthesis of pharmaceuticals, agrochemicals, and advanced materials due to its ability to impart specific chemical properties and structural motifs to organic molecules.