Proteins
Protein catabolism is the biochemical process by which proteins are degraded into amino acids and smaller peptides, providing essential building blocks for new protein synthesis, energy production, and nitrogen homeostasis.
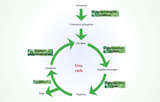
Overview of Pathways
Protein breakdown begins extracellularly via digestive proteases (pepsin, trypsin, chymotrypsin) in the stomach and small intestine, yielding free amino acids for absorption into the bloodstream. Intracellularly, the ubiquitin-proteasome system (UPS) targets misfolded or regulatory proteins: E1/E2/E3 ligases tag substrates with ubiquitin chains, which 26S proteasomes hydrolyze into oligopeptides (7-9 residues) for cytosolic aminopeptidases. Lysosomal autophagy handles bulk degradation, engulfing organelles via autophagosomes fused with lysosomes containing cathepsins.
Amino Acid Fate
Amino groups undergo transamination (ALT/AST enzymes, PLP cofactor) to α-ketoglutarate, forming glutamate, then oxidative deamination via glutamate dehydrogenase yields ammonia (urea cycle) and α-ketoacids. Glucogenic amino acids (e.g., Ala → pyruvate → TCA) fuel gluconeogenesis; ketogenic ones (Leu, Lys → acetyl-CoA → ketones) support brain during starvation. Branched-chain amino acids (BCAAs) catabolize in muscle via BCKDH complex.
Regulation and Significance
mTORC1 activates UPS/autophagy post-feeding; FOXO/AMPK upregulate during nutrient stress. Dysregulation links to cancer (proteasome inhibitors like bortezomib), sarcopenia, and lysosomal storage diseases.
| Pathway | Key Enzymes | Products | Tissues |
|---|---|---|---|
| UPS | Ubiquitin ligases, 26S proteasome | Oligopeptides → AA | All cells, esp. liver/muscle |
| Lysosomal | Cathepsins D/B/L | Free AA | Autophagy, long-lived proteins |
| Digestive | Trypsin, carboxypeptidases | Free AA | Small intestine lumen |
Clinical Relevance
Hypercatabolism in sepsis/cachexia elevates BUN; urea cycle defects (e.g., OTC deficiency) cause hyperammonemia. Protein turnover markers (3-methylhistidine) assess muscle wasting.